In Artinis NIRS blog, you will find the latest trends in (f)NIRS, NIRS studies and applications, tutor from the leaders of near infrared spectroscopy, not to mention detailed insights and tips and tricks for your research!
Search blog post topic
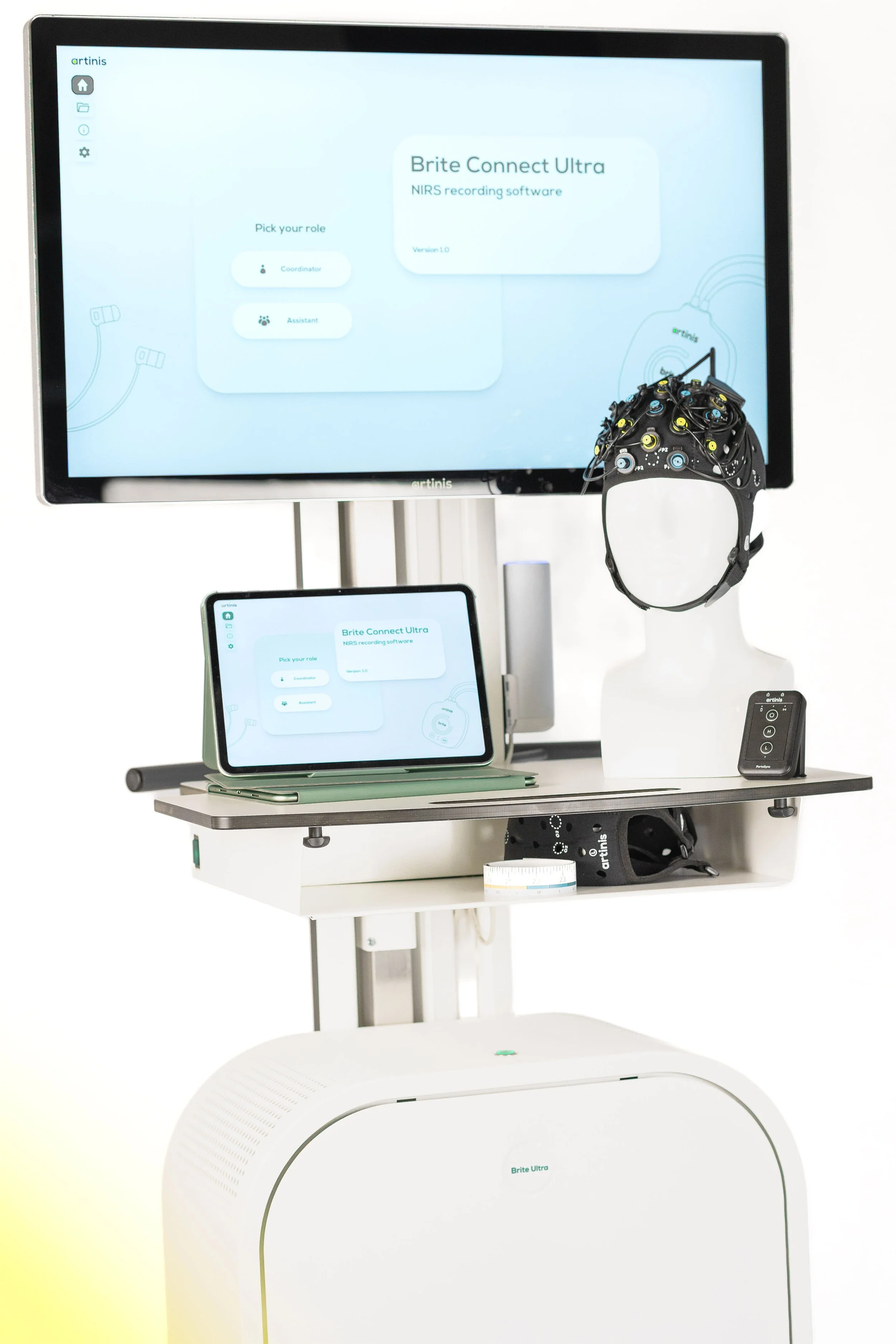
UX Design for Brite Connect Ultra: Making Large-Scale Hyperscanning Truly Scalable
At Artinis, our mission is simple: make imaging easy, so researchers can focus on what matters, their research. Large-scale hyperscanning presented a clear challenge. We answered it the same way we always do: by removing the complexity and making it accessible.

Measuring brain signals in real-time: the potential of using wearable fNIRS in Brain-Computer Interface (BCI)
Given its technical advantages, such as portability, ease of use, and robustness, fNIRS is a suitable neuroimaging tool for application in Brain-Computer Interface (BCI) and Neurofeedback in various settings.
Read this blog to learn more about clinical and daily-life applications of fNIRS-BCI.

The power of portability — Using fNIRS in Naturalistic Settings
Performing research in real-world environments has been gaining importance in neuroscience. Due to its portability, ease of use, and robustness to motion, fNIRS can be perfectly used in naturalistic settings outside the lab. In this blog, we explain the various applications of fNIRS in the real world, highlight study examples, and provide an overview of the fNIRS solutions we offer to enable research in any setting and with any participant.

Brite News: Significantly decrease your setup time with our new Brite
We are happy to announce the release of MKIV of our Brite! Including a novel receiver with increased sensitivity & the latest and enhanced software and accessories, setup time can be significantly decreased with the Brite MKIV —in every subject and/or setting! Learn the new features, novel accessories, and latest updates of our Brite MKIV now!

Integrating muscle NIRS & HD-EMG: Gain advanced muscle insights
Both NIRS and HD-EMG are non-invasive and portable techniques for muscle measurements. Combining these two methodologies can provide complementary information and enable enhanced insights into muscle activity and metabolism in many applications. In this blogpost we explain both techniques and highlight the advantages of combining NIRS and HD-EMG!

Ensuring good fNIRS data quality — Automatic assessment of signal quality
In fNIRS, achieving high data quality is crucial for reliable results, but it can still bear challenges due to hair or skin pigmentation, especially when using fNIRS systems with a higher channel number. In this blog, we present automated signal quality assessment methods that can serve as an efficient solution to detect good and poor data quality in fNIRS measurements.

Which experimental designs to use in fNIRS — Resting-state design
Resting-state design using fNIRS can give information on neural networks and functional brain architecture, and is often used in developmental and clinical studies. In this blogpost, we discuss the characteristics, advantages, and considerations of resting-state experiments and give recommendations for successful fNIRS resting-state studies.

Our Headcap MKII – Improved design for easy and accurate full-head fNIRS measurements
As headcaps are one of the most important accessories in fNIRS experiments, we design our own caps with maximum comfort, fit and quality in mind. To facilitate performance of full-head fNIRS measurements with our MultiBrite devices, we are happy to introduce our Headcap MKII. Read this blogpost to learn more about features and advantages of our new headcap model.

Motion artefacts in (f)NIRS — How to address them
Movement is one of the most common causes for artefacts in neuroimaging tool such as fNIRS. Once motion artefacts are detected, it is important to decide on how to address them to preserve data validity. In this third blogpost of our motion artefact series, we present different methods to correct for motion artefacts and their advantages and disadvantages.

Motion artefacts in (f)NIRS — How to best detect them
Although fNIRS is relatively insensitive for motion artefacts, they cannot be completely prevented. Fortunately, there are various ways to detect motion artefacts. In this second part of our blogpost series on motion artefacts, we explain different methods of detections and elaborate on their advantages and disadvantages.

Artinis and NIRx join forces to accelerate innovation in brain imaging
Artinis Medical Systems, a pioneer in functional Near-Infrared Spectroscopy (fNIRS), is pleased to announce a strategic partnership with NIRx Medical Technologies. With the support of healthcare investor Gilde Healthcare, this collaboration marks a significant milestone in Artinis’ mission to advance neuroscience through accessible, high-impact neuroimaging solutions.

Motion artefacts in (f)NIRS — Not a problem, but not to be overlooked
Due to its portability and ease of use, fNIRS is applied in a variety of research fields, especially in study designs involving movement. Although fNIRS is relatively insensitive to motion artefacts, they still can occur and might influence signal quality. In this first part of our blogpost series on motion artefacts in NIRS, we explain what movement artefacts are and how they can be recognized.
